Tapentadol
 |
|
| Clinical data | |
|---|---|
| Trade names | Nucynta, Palexia |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a610006 |
| Pregnancy category |
|
| Routes of administration |
Oral |
| ATC code | N02AX06 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 32% (oral) |
| Protein binding | 20% |
| Metabolism | Hepatic (mostly via glucuronidation but also by CYP2C9, CYP2C19, CYP2D6) |
| Biological half-life | 4 hours< |
| Excretion | Urine and faeces (1%) |
| Identifiers | |
|
|
| Synonyms | BN-200 CG-5503 R-331333 |
| CAS Number |
175591-09-0 |
| PubChem (CID) | 9838022 |
| IUPHAR/BPS | 7477 |
| ChemSpider |
8013742 |
| UNII |
H8A007M585 |
| ChEMBL |
CHEMBL1201776 |
| ECHA InfoCard | 100.131.247 |
| Chemical and physical data | |
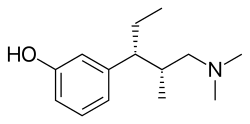
| Formula | C14H23NO |
| Molar mass | 221.339 g/mol |
| 3D model (Jmol) | Interactive image |
| Boiling point | (decomposes) |
|
|
|
|
|
|
|
Tapentadol (brand names: Nucynta, Palexia and Tapal) is a centrally acting opioid analgesic of the benzenoid class with a dual mode of action as an agonist of the μ-opioid receptor and as a norepinephrine reuptake inhibitor (NRI). Analgesia occurs within 32 minutes of oral administration, and lasts for 4–6 hours.
It is similar to tramadol in its dual mechanism of action; namely, its ability to activate the mu opioid receptor and inhibit the reuptake of norepinephrine. Unlike tramadol, it has only weak effects on the reuptake of serotonin and is a significantly more potent opioid with no known active metabolites. Tapentadol is not a pro-drug and therefore does not rely on metabolism to produce its therapeutic effects; this makes it a useful moderate-potency analgesic option for patients who do not respond adequately to more commonly used opioids due to genetic disposition (poor metabolizers of CYP3A4 and CYP2D6), as well as providing a more consistent dosage-response range among the patient population.
Tapentadol general potency is somewhere between that of tramadol and morphine, with an analgesic efficacy comparable to that of oxycodone despite a lower incidence of side effects. It is generally regarded as a weak-moderate strength opioid.
Tapentadol was approved by the US FDA in November 2008, by the TGA of Australia in December 2010 and by the MHRA of the UK in February 2011.1
Tapentadol is used for the treatment of moderate to severe pain for both acute (following injury, surgery, etc.) and chronic musculoskeletal pain. It is also specifically indicated for controlling the pain of diabetic neuropathy when around-the-clock opioid medication is required.
...
Wikipedia
