Triphenylarsine
 |
|
 |
|
| Names | |
|---|---|
|
IUPAC name
Triphenylarsane
|
|
| Other names
Tribenzenidoarsenic
Triphenylarsine |
|
| Identifiers | |
|
3D model (Jmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.009.121 |
| EC Number | 210-032-9 |
|
PubChem CID
|
|
| RTECS number | CH8942500 |
| UN number | 3465 |
|
|
|
|
| Properties | |
| C18H15As | |
| Molar mass | 306.24 g·mol−1 |
| Appearance | Colourless solid |
| Density | 1.395 g cm−3 |
| Melting point | 58 to 61 °C (136 to 142 °F; 331 to 334 K) |
| Boiling point | 373 °C (703 °F; 646 K) at 760 mmHg |
| Insoluble | |
| Solubility | Soluble in ethyl ether, benzene, slightly soluble in ethanol |
| -177.0·10−6 cm3/mol | |
| Structure | |
| Triclinic | |
| Hazards | |
|
EU classification (DSD) (outdated)
|
|
| R-phrases (outdated) | R23/25, R50/53 |
| S-phrases (outdated) | S20/21, S28, S45, S60, S61 |
| Related compounds | |
|
Related organoarsanes
|
Trimethylarsine |
|
Related compounds
|
Triphenylamine |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Triphenylborane
Triphenylphosphine
Triphenylstibine
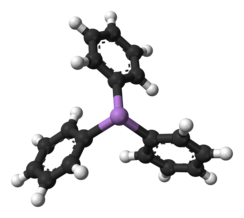
Triphenylarsine is the chemical compound with the formula As(C6H5)3. This organoarsenic compound, often abbreviated AsPh3, is a colorless crystalline solid that is used as a ligand and a reagent in coordination chemistry and organic synthesis. The molecule is pyramidal with As-C distances of 1.942–1.956 Å and C-As-C angles of 99.6–100.5°.
This compound is prepared by the reaction of arsenic trichloride with chlorobenzene using sodium as the reducing agent:
AsPh3 is the precursor to tetraphenylarsonium chloride, [AsPh4]Cl, a popular precipitating agent.
AsPh3 forms complexes with low valent metals that are analogous to the corresponding triphenylphosphine derivatives, such as IrCl(CO)(AsPh3)2 and RhCl(AsPh3)3.
...
Wikipedia
