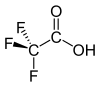
Trifluoroacetic acid
|
|
|||
 |
|||
 |
|||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
Trifluoroacetic acid
|
|||
| Other names
2,2,2-Trifluoroacetic acid
2,2,2-Trifluoroethanoic acid Perfluoroacetic acid Trifluoroethanoic acid TFA |
|||
| Identifiers | |||
|
76-05-1 |
|||
| 3D model (Jmol) | Interactive image | ||
| ChEBI |
CHEBI:45892 |
||
| ChEMBL |
ChEMBL506259 |
||
| ChemSpider |
10239201 |
||
| ECHA InfoCard | 100.000.846 | ||
| PubChem | 6422 | ||
| RTECS number | AJ9625000 | ||
| UNII |
E5R8Z4G708 |
||
|
|||
|
|||
| Properties | |||
| C2HF3O2 | |||
| Molar mass | 114.02 g/mol | ||
| Appearance | colorless liquid | ||
| Density | 1.489 g/cm3, 20 °C | ||
| Melting point | −15.4 °C (4.3 °F; 257.8 K) | ||
| Boiling point | 72.4 °C (162.3 °F; 345.5 K) | ||
| miscible | |||
| Acidity (pKa) | 0.23 | ||
| -43.3·10−6 cm3/mol | |||
| Hazards | |||
| Main hazards | Highly corrosive | ||
| Safety data sheet | External MSDS | ||
| R-phrases | R20 R35 R52/53 | ||
| S-phrases | S9 S26 S27 S28 S45 S61 | ||
| NFPA 704 | |||
| Related compounds | |||
|
Related perfluorinated acids
|
Perfluorooctanoic acid Perfluorononanoic acid |
||
|
Related compounds
|
Acetic acid Trichloroacetic acid |
||
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|||
|
|
|||
| Infobox references | |||
Trifluoroacetic acid (TFA) is an organofluorine compound with the chemical formula CF3CO2H. It is a colorless liquid with a sharp odor similar to vinegar, but stronger in acidity. TFA is an analogue of acetic acid with the three hydrogen atoms replaced by three fluorine atoms. The acidity of TFA is approximately 34,000 times stronger than that of acetic acid due to the electronegativity of the trifluoromethyl group. TFA is widely used in organic chemistry for various purposes.
TFA is prepared industrially by the electrofluorination of acetyl chloride and acetic anhydride, followed by hydrolysis of the resulting trifluoroacetyl fluoride:
Where desired, this compound may be dried by addition of trifluoroacetic anhydride.
An older route to TFA proceeds via the oxidation of 1,1,1-trifluoro-2,3,3-trichloropropene with potassium permanganate. The trifluorotrichloropropene can be prepared by Swarts fluorination of hexachloropropene.
TFA occurs naturally in sea water, but only in small concentrations (<200 ng/L).
TFA is the precursor to many other fluorinated compounds such as trifluoroacetic anhydride, trifluoroperacetic acid, and 2,2,2-trifluoroethanol. It is a reagent used in organic synthesis because of a combination of convenient properties: volatility, solubility in organic solvents, and its strength as an acid. TFA is also less oxidizing than sulfuric acid but more readily available in anhydrous form than many other acids. One complication to its use is that TFA forms an azeotrope with water (b. p. 105 °C).
...
Wikipedia