Droperidol
 |
|
 |
|
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration |
Intravenous, Intramuscular |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Biological half-life | 2.3 hours |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| ECHA InfoCard | 100.008.144 |
| Chemical and physical data | |
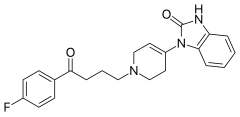
| Formula | C22H22FN3O2 |
| Molar mass | 379.428 g/mol |
| 3D model (Jmol) | |
|
|
|
|
Droperidol /droʊˈpɛrIdɔːl/ (Inapsine, Droleptan, Dridol, Xomolix, Innovar [combination with fentanyl]) is an antidopaminergic drug used as an antiemetic and antipsychotic. Droperidol is also often used for neuroleptanalgesic anesthesia and sedation in intensive-care treatment.
Discovered at Janssen Pharmaceutica in 1961, droperidol is a butyrophenone, and is a potent D2 (dopamine receptor) antagonist with some histamine and serotonin antagonist activity. It has a central antiemetic action and effectively prevents postoperative nausea and vomiting in adults using doses as low as 0.625 mg. It has also been used as an antipsychotic in doses ranging from 5 to 10 mg given as an intramuscular injection, generally in cases of severe agitation in a psychotic patient who is refusing oral medication. Its use in intramuscular sedation has been replaced by intramuscular preparations of haloperidol, midazolam, clonazepam and olanzapine. Some practitioners recommend the use of 0.5 mg to 1 mg intravenously for the treatment of vertigo in an otherwise healthy elderly patients who have not responded to Epley maneuvers.
In 2001, the FDA changed the labeling requirements for droperidol injection to include a Black Box Warning, citing concerns of QT prolongation and torsades de pointes. The evidence for this is disputed, with 9 reported cases of torsades in 30 years and all of those having received doses in excess of 5 mg. QT prolongation is a dose-related effect, and it appears that droperidol is not a significant risk in low doses.
...
Wikipedia
