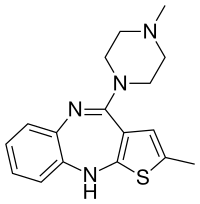
Olanzapine
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Zyprexa (originator), many generics |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601213 |
| License data |
|
| Pregnancy category |
|
| Routes of administration |
oral, intramuscular |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 87% |
| Protein binding | 93% |
| Metabolism | Hepatic (direct glucuronidation and CYP1A2 mediated oxidation) |
| Biological half-life | 33 hours, 51.8 hours (elderly) |
| Excretion | Urine (57%; 7% as unchanged drug), faeces (30%) |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.125.320 |
| Chemical and physical data | |
| Formula | C17H20N4S |
| Molar mass | 312.439 |
| 3D model (Jmol) | |
| Melting point | 195 °C (383 °F) |
| Solubility in water | Practically insoluble in water mg/mL (20 °C) |
|
|
|
|
|
|
|
Olanzapine (originally branded Zyprexa) is an atypical antipsychotic. It is approved by the U.S. Food and Drug Administration (FDA) for the treatment of schizophrenia and bipolar disorder.
Olanzapine is structurally similar to clozapine and quetiapine. It is a dopamine antagonist and is classified as a thienobenzodiazepine.
Olanzapine was first made in the United Kingdom in 1982 by Eli Lilly. The drug became generic in 2011. Sales of Zyprexa in 2008 were $2.2B in the US, and $4.7B worldwide.
The first-line psychiatric treatment for schizophrenia is antipsychotic medication which includes olanzapine. A Cochrane review found, however, that the usefulness for maintenance therapy is difficult to determine as more than half of people in trials quit before the six-week completion date.
National Institute for Health and Care Excellence, the British Association for Psychopharmacology, and the World Federation of Societies for Biological Psychiatry suggest that there is little difference in effectiveness between antipsychotics in prevention of relapse, and recommend that the specific choice of antipsychotic be chosen based on persons preference and side effect profile. The U.S. Agency for Healthcare Research and Quality concludes that olanzapine is not different from haloperidol in the treatment of positive symptoms and general psychopathology, or in overall assessment, but that it is superior for the treatment of negative and depressive symptoms. When trials enrolling only treatment-resistant patients were excluded from the analysis, olanzapine was superior for overall assessment.
A 2013 review of first episode schizophrenia concluded that olanzapine is superior to haloperidol in providing a lower discontinuation rate, and in short-term symptom reduction, response rate, negative symptoms, depression, cognitive function, discontinuation due to poor efficacy, and long-term relapse, but not in positive symptoms or on the Clinical Global Impressions score. In contrast, pooled second generation antipsychotics showed superiority to first generation antipsychotics only against the discontinuation, negative symptoms (with a much larger effect seen among industry- compared to government-sponsored studies), and cognition scores. Olanzapine caused less extrapyramidal side effects, less akathisia, but caused significantly more weight gain, serum cholesterol increase, and triglyceride increase than haloperidol. A 2012 review concluded that among 10 atypical antipsychotics, only clozapine, olanzapine, and risperidone were better than first generation antipsychotics. A 2011 review concluded that neither first- nor second generation antipsychotics produce clinically meaningful changes in Clinical Global Impression scores but found that olanzapine and amisulpride produce larger effects on the PANSS and BPRS batteries than 5 other second generation antipsychotics or pooled first generation antipsychotics.
...
Wikipedia
