Bromine trifluoride
 |
|
 |
|
| Identifiers | |
|---|---|
| ECHA InfoCard | 100.029.211 |
|
PubChem CID
|
|
| Properties | |
| BrF3 | |
| Molar mass | 136.90 g/mol |
| Appearance | straw-coloured liquid hygroscopic |
| Odor | Choking, pungent |
| Density | 2.803 g/cm3 |
| Melting point | 8.77 °C (47.79 °F; 281.92 K) |
| Boiling point | 125.72 °C (258.30 °F; 398.87 K) |
| decomposes violently | |
| Solubility in sulfuric acid | very soluble |
| Structure | |
| T-shaped (C2v) | |
| 1.19 D | |
| Hazards | |
| Main hazards | dangerously sensitive to water, source of HF |
| Safety data sheet |
See: data page http://www.chammascutters.com/en/downloads/Bromine-Trifluoride-MSDS.pdf |
|
EU classification (DSD)
|
|
| NFPA 704 | |
| Related compounds | |
|
Other anions
|
Bromine monochloride |
|
Other cations
|
Chlorine trifluoride Iodine trifluoride |
|
Related compounds
|
Bromine monofluoride Bromine pentafluoride |
| Supplementary data page | |
|
Refractive index (n), Dielectric constant (εr), etc. |
|
|
Thermodynamic
data |
Phase behaviour solid–liquid–gas |
| UV, IR, NMR, MS | |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Bromine trifluoride is an interhalogen compound with the formula BrF3. It is a straw-coloured liquid with a pungent odor. It is soluble in sulfuric acid but explodes on contact with water and organic compounds. It is a powerful fluorinating agent and an ionizing inorganic solvent. It is used to produce uranium hexafluoride (UF6) in the processing and reprocessing of nuclear fuel.
Bromine trifluoride was first described by Paul Lebeau in 1906, who obtained the material by the reaction of bromine with fluorine at 20 °C:
The disproportionation of bromine monofluoride also gives bromine trifluoride:
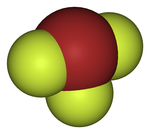
Like ClF3 and IF3, the BrF3molecule is T-shaped and planar. In the VSEPR formalism, the bromine center is assigned two electron pairs. The distance from the bromine each axial fluorine is 1.81 Å and to the equatorial fluorine is 1.72 Å. The angle between an axial fluorine and the equatorial fluorine is slightly smaller than 90° — the 86.2° angle observed is due to the repulsion generated by the electron pairs being greater than that of the Br-F bonds.
BrF3 is a fluorinating agent, but less reactive than ClF3. The liquid is conducting, owing to autoionisation:
Many ionic fluorides dissolve readily in BrF3 forming fluoroanions:
...
Wikipedia

