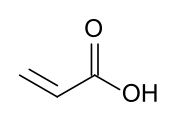
Acrylic acid
 |
|
 |
|
| Names | |
|---|---|
|
Preferred IUPAC name
Prop-2-enoic acid
|
|
| Other names
Acrylic acid
Acroleic acid Ethylenecarboxylic acid Propene acid Propenoic acid Vinylformic acid |
|
| Identifiers | |
|
79-10-7 |
|
| 3D model (Jmol) |
Interactive image Interactive image |
| ChEBI |
CHEBI:18308 |
| ChEMBL |
ChEMBL1213529 |
| ChemSpider |
6333 |
| DrugBank |
DB02579 |
| ECHA InfoCard | 100.001.071 |
| EC Number | 201-177-9 |
| KEGG |
D03397 |
| PubChem | 6581 |
| RTECS number | AS4375000 |
| UNII |
J94PBK7X8S |
|
|
|
|
| Properties | |
| C3H4O2 | |
| Molar mass | 72.06 g·mol−1 |
| Appearance | clear, colorless liquid |
| Odor | acrid |
| Density | 1.051 g/mL |
| Melting point | 14 °C (57 °F; 287 K) |
| Boiling point | 141 °C (286 °F; 414 K) |
| Miscible | |
| Vapor pressure | 3 mmHg |
| Acidity (pKa) | 4.25 |
| Viscosity | 1.3 cP at 20 °C (68 °F) |
| Hazards | |
| Main hazards | Corrosive (C), Dangerous for the environment (N) |
| Safety data sheet | MSDS |
| R-phrases | R10 R20/21/22 R35 R50 |
| S-phrases | S26 S36/37/39 S45 S61 |
| NFPA 704 | |
| Flash point | 68 °C (154 °F; 341 K) |
| 429 °C (804 °F; 702 K) | |
| Explosive limits | 2.4%-8.02% |
| US health exposure limits (NIOSH): | |
|
PEL (Permissible)
|
none |
|
REL (Recommended)
|
TWA 2 ppm (6 mg/m3) [skin] |
|
IDLH (Immediate danger)
|
N.D. |
| Related compounds | |
|
Other anions
|
acrylate |
|
Related carboxylic acids
|
acetic acid propionic acid lactic acid 3-hydroxypropionic acid malonic acid butyric acid crotonic acid |
|
Related compounds
|
allyl alcohol propionaldehyde acrolein methyl acrylate |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Acrylic acid (IUPAC: prop-2-enoic acid) is an organic compound with the formula CH2=CHCOOH. It is the simplest unsaturated carboxylic acid, consisting of a vinyl group connected directly to a carboxylic acid terminus. This colorless liquid has a characteristic acrid or tart smell. It is miscible with water, alcohols, ethers, and chloroform. More than a million tons are produced annually.
Acrylic acid is produced from propylene which is a byproduct of ethylene and gasoline production.
Ethylene can be carboxylated to acrylic acid under supercritical carbon dioxide condition.
Because acrylic acid and its esters have long been valued commercially, many other methods have been developed but most have been abandoned for economic or environmental reasons. An early method was the hydrocarboxylation of acetylene ("Reppe chemistry"):
This method requires nickel carbonyl and high pressures of carbon monoxide. It was once manufactured by the hydrolysis of acrylonitrile which is derived from propene by ammoxidation, but was abandoned because the method cogenerates ammonium derivatives. Other now abandoned precursors to acrylic acid include ethenone and ethylene cyanohydrin.
Dow Chemical Company and a partner, OPX Biotechnologies, are investigating using fermented sugar to produce 3-hydroxypropionic acid (3HP), an acrylic acid precursor. The goal is to reduce greenhouse gas emissions.
...
Wikipedia

