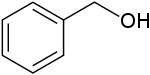
Benzyl alcohol
 |
|
 |
|
| Names | |
|---|---|
|
IUPAC name
Phenylmethanol
|
|
| Other names
Phenylcarbinol
|
|
| Identifiers | |
|
100-51-6 |
|
| 3D model (Jmol) |
Interactive image Interactive image |
| ChEBI |
CHEBI:17987 |
| ChEMBL |
ChEMBL720 |
| ChemSpider |
13860335 |
| ECHA InfoCard | 100.002.600 |
| EC Number | 202-859-9 |
| E number | E1519 (additional chemicals) |
| KEGG |
D00077 |
| PubChem | 244 |
| RTECS number | DN3150000 |
| UNII |
LKG8494WBH |
|
|
|
|
| Properties | |
| C7H8O | |
| Molar mass | 108.14 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Slightly aromatic |
| Density | 1.044 g cm−3 |
| Melting point | −15.2 °C (4.6 °F; 257.9 K) |
| Boiling point | 205.3 °C (401.5 °F; 478.4 K) |
| 3.50 g/100 mL (20 °C) 4.29 g/100 mL (25 °C) |
|
| Solubility in other solvents | Soluble in benzene, methanol, chloroform, ethanol, ether, acetone |
| log P | 1.10 |
| Vapor pressure | 0.18 kPa (60 °C) |
| Acidity (pKa) | 15.40 |
| -71.83·10−6 cm3/mol | |
|
Refractive index (nD)
|
1.5396 |
| Viscosity | 5.474 cP |
| 1.67 D | |
| Thermochemistry | |
|
Std molar
entropy (S |
217.8 J/K mol |
|
Std enthalpy of
formation (ΔfH |
-352 kJ/mol |
| Pharmacology | |
| P03AX06 (WHO) | |
| Hazards | |
| Safety data sheet | External MSDS |
| NFPA 704 | |
| Flash point | 93 °C (199 °F; 366 K) |
| 436 °C (817 °F; 709 K) | |
| Explosive limits | 1.3–13% |
| Lethal dose or concentration (LD, LC): | |
|
LD50 (median dose)
|
1250 mg/kg (rat, oral) |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Benzyl alcohol is an aromatic alcohol with the formula C6H5CH2OH. The benzyl group is often abbreviated "Bn" (not to be confused with "Bz" which is used for benzoyl), thus benzyl alcohol is denoted as BnOH. Benzyl alcohol is a colorless liquid with a mild pleasant aromatic odor. It is a useful solvent due to its polarity, low toxicity, and low vapor pressure. Benzyl alcohol has moderate solubility in water (4 g/100 mL) and miscible in alcohols and diethyl ether. The anion produced by deprotonation of the alcohol group is known as benzylate or benzyloxide.
Benzyl alcohol is produced naturally by many plants and is commonly found in fruits and teas. It is also found in a variety of essential oils including jasmine, hyacinth, and ylang-ylang. It is also found in castoreum from the castor sacs of beavers.
Benzyl alcohol is prepared by the hydrolysis of benzyl chloride using sodium hydroxide:
It can also be prepared via a Grignard reaction by reacting phenylmagnesium bromide (C6H5MgBr) with formaldehyde, followed by acidification.
Like most alcohols, it reacts with carboxylic acids to form esters. In organic synthesis, benzyl esters are popular protecting groups because they can be removed by mild hydrogenolysis.
...
Wikipedia

