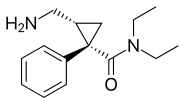
Levomilnacipran
 |
|
| Clinical data | |
|---|---|
| Trade names | Fetzima |
| Pregnancy category |
|
| Routes of administration |
By mouth (capsules) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 92% |
| Protein binding | 22% |
| Metabolism | Hepatic (primarily by CYP3A4) |
| Biological half-life | 12 hours |
| Excretion | Renal |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C15H22N2O |
| Molar mass | 246.348 g/mol |
| 3D model (Jmol) | |
|
|
|
|
|
|
|
Levomilnacipran (brand name Fetzima) is an antidepressant approved in the United States for the treatment of major depressive disorder (MDD) in adults. It was developed by Forest Laboratories and Pierre Fabre Group, and was approved by the Food and Drug Administration in July 2013. Levomilnacipran is the levo- enantiomer of milnacipran, and has similar effects and pharmacology, acting as a serotonin-norepinephrine reuptake inhibitor (SNRI).
The FDA approved levomilnacipran in July 2013 based on the results of one 10-week phase II and four 8-week phase III clinical trials. Four of the five trials demonstrated a statistically significant superiority to placebo as measured by the Montgomery–Åsberg Depression Rating Scale. Superiority to placebo was also demonstrated by improvement in the Sheehan Disability Scale. Side effects seen more often than with placebo included nausea, dizziness, sweating, constipation, insomnia, increased heart rate and blood pressure, urinary hesitancy, erectile dysfunction and delayed ejaculation in males, vomiting, tachycardia, and palpitations.
...
Wikipedia
