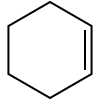
Cyclohexene
|
|
|||
|
|
|||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
Cyclohexene
|
|||
| Other names
Tetrahydrobenzene, 1,2,3,4-Tetrahydrobenzene, Benzenetetrahydride, Cyclohex-1-ene, Hexanaphthylene, UN 2256
|
|||
| Identifiers | |||
|
110-83-8 |
|||
| 3D model (Jmol) | Interactive image | ||
| ChEBI |
CHEBI:36404 |
||
| ChEMBL |
ChEMBL16396 |
||
| ChemSpider |
7788 |
||
| ECHA InfoCard | 100.003.462 | ||
| EC Number | 203-807-8 | ||
| PubChem | 8079 | ||
| RTECS number | GW2500000 | ||
|
|||
|
|||
| Properties | |||
| C6H10 | |||
| Molar mass | 82.143 g/mol | ||
| Appearance | colorless liquid | ||
| Odor | sweet | ||
| Density | 0.8110 g/cm3 | ||
| Melting point | −103.5 °C (−154.3 °F; 169.7 K) | ||
| Boiling point | 82.98 °C (181.36 °F; 356.13 K) | ||
| insoluble in water | |||
| Solubility | miscible with organic solvents | ||
| Vapor pressure | 8.93 kPa (20 °C)
11.9 kPa (25 °C) |
||
|
Henry's law
constant (kH) |
0.022 mol·kg−1·bar−1 | ||
| -57.5·10−6 cm3/mol | |||
|
Refractive index (nD)
|
1.4465 | ||
| Hazards | |||
| Safety data sheet | External MSDS | ||
| R-phrases | R11, R19, R21/22 | ||
| S-phrases | S16, S23, S24, S25, S33 | ||
| NFPA 704 | |||
| Flash point | −12 °C (10 °F; 261 K) | ||
| 244 °C (471 °F; 517 K) | |||
| Explosive limits | 0.8–5 % | ||
| Lethal dose or concentration (LD, LC): | |||
|
LD50 (median dose)
|
1946 mg/kg (oral, rat) | ||
|
LCLo (lowest published)
|
13,196 ppm (mouse, 2 hr) | ||
| US health exposure limits (NIOSH): | |||
|
PEL (Permissible)
|
TWA 300 ppm (1015 mg/m3) | ||
|
REL (Recommended)
|
TWA 300 ppm (1015 mg/m3) | ||
|
IDLH (Immediate danger)
|
2000 ppm | ||
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|||
|
|
|||
| Infobox references | |||
11.9 kPa (25 °C)
Cyclohexene is a hydrocarbon with the formula C6H10. This cycloalkene is a colorless liquid with a sharp smell. It is an intermediate in various industrial processes. Cyclohexene is not very stable upon long term storage with exposure to light and air because it forms peroxides.
Cyclohexene is produced by the partial hydrogenation of benzene, a process developed by the Asahi Chemical company. It is converted to cyclohexanol, which is dehydrogenated to give cyclohexanone, a precursor to caprolactam. Cyclohexene is also a precursor to adipic acid, maleic acid, dicyclohexyladipate, and cyclohexene oxide. Furthermore, it is used as a solvent.
A common experiment for beginning organic chemistry students is the acid-catalyzed dehydration of cyclohexanol with distillative removal of the resulting cyclohexene from the reaction mixture:
A green chemistry experiment is the oxidative cleavage of cyclohexene to form adipic acid. Hydrogen peroxide is used as the oxidant, in the presence of a tungsten catalyst.
Cyclohexene is most stable in a half-chair conformation, unlike the preference for a chair for of cyclohexane. One basis for the cyclohexane conformational preference for a chair is that it allows each bond of the ring to adopt a staggered conformation. For cyclohexene, however, the alkene is planar, equivalent to an eclipsed conformation at that bond.
...
Wikipedia