Carbon monoxide toxicity
| Carbon monoxide poisoning | |
|---|---|
 |
|
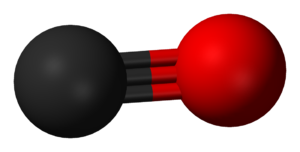
| Carbon monoxide | |
| Classification and external resources | |
| Specialty | Toxicology |
| ICD-10 | T58 |
| ICD-9-CM | 986 |
| eMedicine | article/819987 |
| Patient UK | Carbon monoxide poisoning |
| MeSH | D002249 |
Carbon monoxide poisoning occurs after breathing in too much carbon monoxide (CO). Symptoms of mild acute poisoning include lightheadedness, confusion, headache, feeling like the world is spinning, and flu-like effects. Larger exposures can lead to toxicity of the central nervous system and heart, and death. After acute poisoning, long-term problems may occur. Carbon monoxide can also have negative effects on a baby if exposed during pregnancy. Chronic exposure to low levels of carbon monoxide can lead to depression, confusion, and memory loss.
Carbon monoxide is a toxic gas, but, being colorless, odorless, tasteless, and initially non-irritating, it is difficult for people to detect. Carbon monoxide is a product of incomplete combustion of organic matter. It is often produced by motor vehicles that run on gasoline, diesel, methane, or other carbon-based fuels and from tools, gas heaters, and cooking equipment that are powered by carbon-based fuels such as propane, butane and charcoal. Exposure at 100 ppm or greater can be dangerous to human health. Carbon monoxide mainly causes adverse effects by combining with hemoglobin to form carboxyhemoglobin (HbCO) in the blood. This prevents hemoglobin from carrying oxygen to the tissues. Additionally, myoglobin and are thought to be adversely affected. Carboxyhemoglobin can revert to hemoglobin, but the recovery takes time as the HbCO complex is fairly stable.
...
Wikipedia
