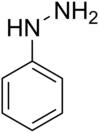
Phenyl hydrazine
 |
|||
|
|
|||
| Names | |||
|---|---|---|---|
|
IUPAC name
Phenylhydrazine
|
|||
| Other names
Hydrazinobenzene
|
|||
| Identifiers | |||
|
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.002.612 | ||
| KEGG | |||
|
PubChem CID
|
|||
| UNII | |||
|
|||
|
|||
| Properties | |||
| C6H8N2 | |||
| Molar mass | 108.14 g/mol | ||
| Appearance | Colorless to pale-yellow liquid or solid | ||
| Odor | faint, aromatic | ||
| Density | 1.0978 g/cm3 | ||
| Melting point | 19.5 °C (67.1 °F; 292.6 K) (24 °C for hemihydrate) | ||
| Boiling point | 243.5 °C (470.3 °F; 516.6 K) (decomposition) | ||
| Vapor pressure | 0.04 mmHg (25°C) | ||
| -67.82·10−6 cm3/mol | |||
| Hazards | |||
| NFPA 704 | |||
| Flash point | 88 °C; 190 °F; 361 K | ||
| Lethal dose or concentration (LD, LC): | |||
|
LD50 (median dose)
|
188 mg/kg (rat, oral) 175 mg/kg (mouse, oral) 80 mg/kg (rabbit, oral) 80 mg/kg (guinea pig, oral) 200-250 mg/kg (dog, oral) |
||
| US health exposure limits (NIOSH): | |||
|
PEL (Permissible)
|
TWA 5 ppm (22 mg/m3) [skin] | ||
|
REL (Recommended)
|
Ca C 0.14 ppm (0.6 mg/m3) [2-hr] [skin] | ||
|
IDLH (Immediate danger)
|
Ca [15 ppm] | ||
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|||
|
|
|||
| Infobox references | |||
Phenylhydrazine is the chemical compound with the formula C6H5NHNH2. It is often abbreviated as PhNHNH2.
Phenylhydrazine forms monoclinic prisms that melt to an oil around room temperature which may turn yellow to dark red upon exposure to air. Phenylhydrazine is miscible with ethanol, diethyl ether, chloroform, and benzene. It is sparingly soluble in water.
Phenylhydrazine is prepared by oxidizing aniline with sodium nitrite in the presence of hydrogen chloride to form the diazonium salt, which is subsequently reduced using sodium sulfite in the presence of sodium hydroxide to form the final product.
Phenylhydrazine was the first hydrazine derivative characterized, reported by Emil Fischer in 1875. He prepared it by reduction of a phenyl diazonium salt using sulfite salts. Fischer used phenylhydrazine to characterize sugars via formation of hydrazones with the sugar aldehyde. He also demonstrated in this first paper many of the key properties recognized for hydrazines.
Phenyl-hydrazine is used to prepare indoles via the Fischer Indole Synthesis, which are intermediates in the synthesis of various dyes and pharmaceuticals.
Phenylhydrazine is used to form phenylhydrazones of natural mixtures of simple sugars in order to render the differing sugars easily separable from each other.
...
Wikipedia



