Oxcarbazepine
 |
|
 |
|
| Clinical data | |
|---|---|
| Pronunciation | /ɒks.kɑːrˈbæz.ᵻˌpiːn/ |
| Trade names | Trileptal |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601245 |
| Pregnancy category |
|
| Routes of administration |
By mouth (tablets or liquid) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | >95% |
| Metabolism | Liver (cytosolic enzymes and glucuronic acid) |
| Biological half-life | 1–5 hours (healthy adults) |
| Excretion | Kidney (<1%) |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| ECHA InfoCard | 100.044.702 |
| Chemical and physical data | |
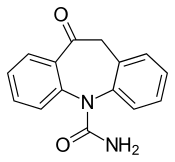
| Formula | C15H12N2O2 |
| Molar mass | 252.268 g/mol |
| 3D model (Jmol) | |
|
|
|
|
Oxcarbazepine is an anticonvulsant drug primarily used in the treatment of epilepsy. There is some evidence for oxcarbazepine as a mood-stabilizing agent and thus, it can be used as add-on therapy for bipolar disorder in patients that have failed or are unable to tolerate approved treatments.
Common side effects include nausea, vomiting, dizziness, drowsiness, headache, double vision and trouble with walking. Although not common, anaphylaxis may occur. Due to its structural similarities to carbamazepine there is approximately a 25–30% chance of cross-reactivity between the two medications.
Oxcarbazepine is marketed as Trileptal by Novartis and available in some countries as a generic drug. There is also an extended-release formulation marketed as Oxtellar XR by Supernus Pharmaceuticals.
Oxcarbazepine is an anticonvulsant used to reduce the occurrence of epileptic episodes, and is not intended to cure epilepsy. Oxcarbazepine is used alone or in combination with other medications for the treatment of focal (partial) seizures in adults. In pediatric populations, it can be used by itself for the treatment of partial seizures for children 4 years and older, or in combination with other medications for children 2 years and older.
In addition, oxcarbazepine has been shown to improve mood and reduce anxiety; therefore, it is a potential option for add-on therapy in the treatment of bipolar disorder.
Oxcarbazepine is listed as Pregnancy Category C.
Currently, there is limited data analyzing the impact of oxcarbazepine on a human fetus. Animal studies have shown an increased incidence of fetal abnormalities in pregnant rats and rabbits treated with oxcarbazepine, during pregnancy. In addition, oxcarbazepine is structurally similar to carbamazepine (pregnancy category: D) which is considered to be teratogenic in humans. Oxcarbazepine should only be used in pregnancy if the benefits justify the risks.
Pregnant persons on oxcarbazepine should be closely monitored, as plasma levels of the active metabolite (MHD) has been shown to potentially decrease during pregnancy.
Oxcarbazepine and MHD are both present in human breast milk and thus, some of the active drug can be transferred to a nursing infant. When considering whether to continue this medication in nursing mothers, the impact of the drug's side effect profile on the infant, should be weighed against its anti-epileptic benefit for the mother.
...
Wikipedia
