Nitrosonium
 |
|
| Names | |
|---|---|
|
Systematic IUPAC name
Oxidonitrogen(1+)
|
|
| Other names
Nitrosonium
Iminooxidanium |
|
| Identifiers | |
| 14452-93-8 | |
| 3D model (Jmol) | Interactive image |
| Abbreviations | NO(+) |
| ChEBI | CHEBI:29120 |
| ChemSpider | 76569 |
| 456 | |
| PubChem | 84878 |
|
|
|
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
| Infobox references | |
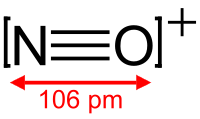
The nitrosonium ion is NO+, in which the nitrogen atom is bonded to an oxygen atom with a bond order of 3, and the overall diatomic species bears a positive charge. This ion is usually obtained as the following salts: NOClO4, NOSO4H (nitrosylsulfuric acid, more descriptively written ONSO3OH), and NOBF4. The ClO−
4 and BF−
4 salts are slightly soluble in CH3CN. NOBF4 can be purified by sublimation at 200–250 °C and 0.01 mmHg (1.3 Pa).
NO+ is isoelectronic with CO, CN− and N2. It arises via protonation of nitrous acid:
NO+ reacts readily with water to form nitrous acid:
For this reason, NOBF4 must be protected from water or even moist air. With base, the reaction generates nitrite:
NO+ reacts with aryl amines, ArNH2, to give diazonium salts, ArN+
2. The resulting diazonium group is easily displaced (unlike the amino group) by a variety of nucleophiles.
NO+, e.g. as NOBF4, is a strong oxidizing agent:
NOBF4 is a convenient oxidant because the byproduct NO is a gas, which can be swept from the reaction using a stream of N2. Upon contact with air, NO forms NO2, which can cause secondary reactions if it is not removed. NO2 is readily detectable by its characteristic orange color.
...
Wikipedia
