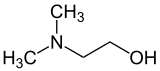
Dimethylaminoethanol
 |
|
 |
|
| Names | |
|---|---|
|
IUPAC name
2-(Dimethylamino)ethanol
|
|
Other names
|
|
| Identifiers | |
|
3D model (Jmol)
|
|
| Abbreviations | DMAE, DMEA |
| 1209235 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.003.221 |
| EC Number | 203-542-8 |
| KEGG | |
| MeSH | Deanol |
|
PubChem CID
|
|
| RTECS number | KK6125000 |
| UNII | |
| UN number | 2051 |
|
|
|
|
| Properties | |
| C4H11NO | |
| Molar mass | 89.14 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Fishy, ammoniacal |
| Density | 890 mg mL−1 |
| Melting point | −59.00 °C; −74.20 °F; 214.15 K |
| Boiling point | 134.1 °C; 273.3 °F; 407.2 K |
| log P | −0.25 |
| Vapor pressure | 816 Pa (at 20 °C) |
| Acidity (pKa) | 9.23 (at 20 °C) |
| Basicity (pKb) | 4.77 (at 20 °C) |
|
Refractive index (nD)
|
1.4294 |
| Pharmacology | |
| N06BX04 (WHO) | |
| Hazards | |
| GHS pictograms |
  
|
| GHS signal word | DANGER |
| H226, H302, H312, H314, H332 | |
| P280, P305+351+338, P310 | |
|
EU classification (DSD)
|
|
| R-phrases | R10, R20/21/22, R34 |
| S-phrases | (S1/2), S25, S26, S36/37/39, S45 |
| Flash point | 39 °C (102 °F; 312 K) |
| Explosive limits | 1.4–12.2% |
| Lethal dose or concentration (LD, LC): | |
|
LD50 (median dose)
|
|
| Related compounds | |
|
Related alkanols
|
|
|
Related compounds
|
Diethylhydroxylamine |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Dimethylaminoethanol and dimethylethanolamine (DMAE and DMEA respectively), with deanol, are common names for 2-(dimethylamino)ethanol.
This compound has tertiary amine and primary alcohol groups as functional groups. The alcohol functionality, in combination with the amine and its low molecular weight imparts water solubility on the compound, and makes ester derivatives possible; the amine functionality gives the compound its alkaline character, and allows for formulation of salts of the compound.
Although preliminary, it seems that DMAE may aggravate or induce the formation of neural tube defects, as evidenced by in vitro studies on mouse embryonic cells. Under normal conditions, rat embryos uptake choline and convert it into phosphatidylcholine(PC) as an active substrate. DMAE supplementation seems to replace choline usage when it is present due to higher affinity and competition but fails to convert into PC due to an apparent lack of expression of the enzyme phosphatidylethanolamine methyltransferase (PeMT) in embryos. This enzyme is absolutely necessary for the conversion of Phosphatidylethanolamine into PC.
Essentially, DMAE competitively inhibits Choline uptake during the first few days of neural tube formation (1–10 days after impregnation), but the embryo is not yet able to use DMAE as well as an adult due to an immature CDP-choline metabolic pathway and underexpression of key enzymes.
These mechanisms should not occur in adult cells due to the activity of the PeMT enzyme and a mature CDP-choline metabolic enzyme pathway.
Its characteristics arise from its chemical features as well as from its overall structural similarity to chemical compounds involved in primary metabolism and neurophysiology.
According to WebMD:
"Deanol is a chemical that is involved in a series of reactions that form acetylcholine, a chemical that is found in the brain and other areas of the body. Acetylcholine is a "neurotransmitter" that helps nerve cells communicate.
Deanol is used for treating attention deficit-hyperactivity disorder (ADHD), Alzheimer's disease, autism, and a movement disorder called tardive dyskinesia. It is also used for improving memory and mood; boosting thinking skills and intelligence; and increasing physical energy, oxygen efficiency, athletic performance, and muscle reflexes. It is also used for preventing aging or liver spots, improving red blood cell function, and extending life span.
...
Wikipedia
