BDTH2
 |
|
 |
|
| Names | |
|---|---|
|
IUPAC name
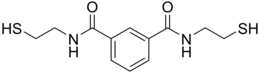
N,N′-bis(2-mercaptoethyl)isophthalamide
|
|
| Other names
BDET; BDTH2; BDETH2; N,N′-Bis(2-mercaptoethyl)-1,3-benzenedicarboxamide; N1,N3-bis(2-mercaptoethyl)isophthalamide; NBMI;
|
|
| Identifiers | |
|
3D model (Jmol)
|
|
| ChemSpider | |
| MeSH | 1,3-benzenediamidoethanethiol |
|
PubChem CID
|
|
|
|
|
|
| Properties | |
| C12H16N2O2S2 | |
| Molar mass | 284.39 g·mol−1 |
| Density | 1.23 g/mL |
| Melting point | 132 to 135 °C (270 to 275 °F; 405 to 408 K) |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
BDTH2 (also called BDET and BDETH2; trade names B9, MetX and OSR#1) is an organosulfur compound that is used as a chelation agent. It is a colourless solid. The molecule consists of two thiol groups and linked via a pair of amide groups.
The compound was reported in about 1994 after a search for chelating agents selective for mercury. It was licensed in 2006 to CTI Science with the long-term goal of using BDTH2 in mercury poisoning. This compound is prepared by treating isophthaloyl dichloride with two equiv of cysteamine:
BDTH2 can be used to chelate heavy metals like lead, cadmium, copper, manganese, zinc, iron, and mercury from ground water, coal tailings, gold ore, waste water of battery-recycling plants, and contaminated soil.
BDTH2 appears to bind mercury more strongly than do other chelators. The mercury-BDT complex does not break down even at high pH and in the presence of cyanides, as in waste water of gold mines. The particular stability of the mercury bond can be attributed to the linear position of the two thiols. The company Covalent Research Technologies had investigated BDTH2 for the removal of mercury from flue gas without success.
Animal experiments with inorganic mercury showed, that BDTH2 effectively binds mercury in the body, and the resulting mercury derivative is excreted in the feces. Experimental animals showed no signs of poisoning. It is unclear, how the BDTH2-mercury-chelate behaves in the long term. BDTH2 is lipophilic, as opposed to DMPS and DMSA and thus can cross lipid membranes including the blood-brain-barrier and enters the bone marrow. In animal experiments, the amount of mercury in brain tissue was not increased, but also not decreased(can anyone provide a reference for the material/data/research from which this conclusion was drawn?). However, there are indications that the BDTH2-mercury-compound moves into adipose tissue. It is unknown, how BDTH2 works with methyl-mercury.
...
Wikipedia
