Aprotinin
 |
|
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Dependence liability |
None |
| Routes of administration |
Intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 100% (intravenous) |
| Identifiers | |
|
|
| Synonyms | Trasylol, bovine pancreatic trypsin inhibitor |
| CAS Number | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEMBL | |
| ECHA InfoCard | 100.029.983 |
| Chemical and physical data | |
| Formula | C284H432N84O79S7 |
| Molar mass | 6511.51 g/mol |
|
|
|
| Aprotinin | |
|---|---|
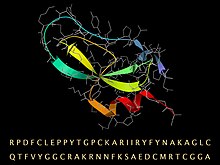
BPTI sequence, with its folded 3D structure represented by a ribbon for the secondary structure and a stick model (gray) for the backbone and sidechains.
|
|
| Identifiers | |
| Organism | |
| Symbol | PTI |
| Entrez | 404172 |
| PDB | 4PTI More structures |
| RefSeq (mRNA) | NM_001001554 |
| RefSeq (Prot) | NP_001001554 |
| UniProt | P00974 |
| Other data | |
| Chromosome | 13: 75.02 - 75.03 Mb |
The drug aprotinin (Trasylol, previously Bayer and now Nordic Group pharmaceuticals), is the small protein bovine pancreatic trypsin inhibitor (BPTI), an antifibrinolytic molecule that inhibits trypsin and related proteolytic enzymes. Under the trade name Trasylol, aprotinin was used as a medication administered by injection to reduce bleeding during complex surgery, such as heart and liver surgery. Its main effect is the slowing down of fibrinolysis, the process that leads to the breakdown of blood clots. The aim in its use was to decrease the need for blood transfusions during surgery, as well as end-organ damage due to hypotension (low blood pressure) as a result of marked blood loss. The drug was temporarily withdrawn worldwide in 2007 after studies suggested that its use increased the risk of complications or death; this was confirmed by follow-up studies. Trasylol sales were suspended in May 2008, except for very restricted research use. In February 2012 the European Medicines Agency (EMA) scientific committee reverted its previous standpoint regarding aprotinin, and has recommended that the suspension be lifted. Nordic became distributor of aprotinin in 2012.
Aprotinin is a monomeric (single-chain) globular polypeptide derived from bovine lung tissue. It has a molecular weight of 6512 and consists of 16 different amino acid types arranged in a chain 58 residues long that folds into a stable, compact tertiary structure of the 'small SS-rich" type, containing 3 disulfides, a twisted β-hairpin and a C-terminal α-helix.
The amino acid sequence for bovine BPTI is RPDFC LEPPY TGPCK ARIIR YFYNA KAGLC QTFVY GGCRA KRNNF KSAED CMRTC GGA. There are 10 positively charged lysine (K) and arginine (R) side chains and only 4 negative aspartate (D) and glutamates (E), making the protein strongly basic, which accounts for the basic in its name. (Because of the usual source organism, BPTI is sometimes referred to as bovine pancreatic trypsin inhibitor.)
...
Wikipedia
