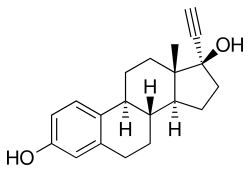
17α-ethinylestradiol
 |
|
 |
|
| Clinical data | |
|---|---|
| Pronunciation | /ˌɛθɪnɪlˌiːstrəˈdaɪ.əl/ |
| Trade names | Estinyl, others |
| Synonyms | 17α-Ethynylestradiol; 17α-Ethynylestra-1,3,5(10)-triene-3,17β-diol; NSC-10973 |
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a604032 |
| Pregnancy category |
|
| Routes of administration |
• By mouth (tablet) • Transdermal (patch) • Vaginal (ring) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 38–48% |
| Protein binding | 97–98% (to albumin; does not bind to SHBG) |
| Metabolism | Liver (primarily CYP3A4) |
| Biological half-life | 7–36 hours |
| Excretion | Feces: 62% Urine: 38% |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.000.311 |
| Chemical and physical data | |
| Formula | C20H24O2 |
| Molar mass | 296.403 g/mol |
| 3D model (JSmol) | |
|
|
|
|
|
|
|
Ethinylestradiol (EE) is an orally active estrogen and a synthetic derivative of estradiol, a steroid hormone and the major endogenous estrogen in humans. EE is used as a medication and is a component of almost all formulations of combined oral contraceptive (COC) pills, being nearly the exclusive estrogen used for this purpose.
EE was patented in 1935 and came into medical use in 1943. It started being used in oral contraceptives in 1964.
Under the brand name Estinyl, EE was formerly used for hormone replacement therapy in menopause and the treatment of female hypogonadism, loss of menstruation, dysmenorrhea, acne, prostate cancer, and breast cancer. However, in more recent times, EE is mainly used in COCs. In contraception, due to concerns of unopposed estrogen action and the possible increased risk of endometrial cancer that accompanies this, EE is formulated in combination with progestins. EE is little used in menopausal hormone replacement therapy.
EE has been used at very high dosages (1–2 mg/day) in the treatment of prostate cancer.
EE should be avoided in women with a history of or known susceptibility to thrombosis (blood clots), particularly venous thromboembolism (VTE).
...
Wikipedia
