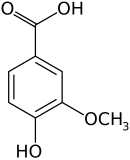
Vanillic acid
|
|
|||
| Names | |||
|---|---|---|---|
|
IUPAC name
4-Hydroxy-3-methoxybenzoic acid
|
|||
| Other names
4-Hydroxy-m-anisic acid, Vanillate
|
|||
| Identifiers | |||
|
121-34-6 |
|||
| 3D model (Jmol) | Interactive image | ||
| ChEBI |
CHEBI:30816 |
||
| ChEMBL |
ChEMBL120568 |
||
| ChemSpider |
8155 |
||
| ECHA InfoCard | 100.004.061 | ||
| PubChem | 8468 | ||
|
|||
|
|||
| Properties | |||
| C8H8O4 | |||
| Molar mass | 168.15 g·mol−1 | ||
| Appearance | White to light yellow powder or crystals | ||
| Melting point | 210 to 213 °C (410 to 415 °F; 483 to 486 K) | ||
| Hazards | |||
| NFPA 704 | |||
| Related compounds | |||
|
Related compounds
|
Vanillin, vanillyl alcohol | ||
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|||
|
|
|||
| Infobox references | |||
Vanillic acid (4-hydroxy-3-methoxybenzoic acid) is a dihydroxybenzoic acid derivative used as a flavoring agent. It is an oxidized form of vanillin. It is also an intermediate in the production of vanillin from ferulic acid.
The highest amount of vanillic acid in plants known so far is found in the root of Angelica sinensis, an herb indigenous to China, which is used in traditional Chinese medicine.
Açaí oil, obtained from the fruit of the açaí palm (Euterpe oleracea), is rich in vanillic acid (1,616 ± 94 mg/kg).
It is one of the main natural phenols in argan oil.
It is also found in wine and vinegar.
Vanillic acid is one of the main catechins metabolites found in humans after consumption of green tea infusions.
Vanillic acid is used in the synthesis of the analeptic drug etamivan.
...
Wikipedia