Tetrakis(triphenylphosphine)palladium
 |
|
 |
|
| Names | |
|---|---|
|
IUPAC name
Tetrakis(triphenylphosphane)palladium(0)
|
|
| Other names
TPP palladium(0)
|
|
| Identifiers | |
| ECHA InfoCard | 100.034.609 |
|
PubChem CID
|
|
| RTECS number | Unregistered |
| Properties | |
| C72H60P4Pd | |
| Molar mass | 1,155.59 g·mol−1 |
| Appearance | Bright yellow crystals |
| Melting point | decomposes around 115 °C |
| Insoluble | |
| Structure | |
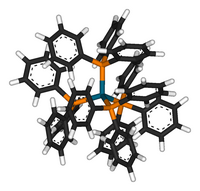
| four triphenylphosphine monodentate ligands attached to a central Pd(0) atom in a tetrahedral geometry |
|
| tetrahedral | |
| 0 D | |
| Hazards | |
| R-phrases (outdated) | n/a |
| S-phrases (outdated) | S22, S24/25 |
| NFPA 704 | |
| Related compounds | |
|
Related complexes
|
chlorotris(triphenylphosphine)rhodium(I) tris(dibenzylideneacetone)dipalladium(0) |
|
Related compounds
|
triphenylphosphine |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Tetrakis(triphenylphosphine)palladium(0) (sometimes called quatrotriphenylphosphine) is the chemical compound Pd[P(C6H5)3]4, often abbreviated Pd(PPh3)4, or even PdP4. It is a bright yellow crystalline solid that becomes brown upon decomposition in air.
The four phosphorus atoms are at the corners of a tetrahedron surrounding the palladium(0) center. This structure is typical for four-coordinate 18 e− complexes. The corresponding complexes Ni(PPh3)4 and Pt(PPh3)4 are also well known. Such complexes reversibly dissociate PPh3 ligands in solution, so reactions attributed to Pd(PPh3)4 often in fact arise from Pd(PPh3)3 or even Pd(PPh3)2.
Tetrakis(triphenylphosphine)palladium(0) was first prepared by Lamberto Malatesta et al. in the 1950s by reduction of sodium chloropalladate with hydrazine in the presence of the phosphine. It is commercially available, but can be prepared in two steps from Pd(II) precursors:
Both steps may be carried out in a one-pot reaction, without isolating and purifying the PdCl2(PPh3)2 intermediate.Reductants other than hydrazine can be employed. The compound is sensitive to air, but can be purified by washing with methanol to give the desired yellow powder. It is usually stored cold under argon.
Pd(PPh3)4 is widely used as a catalyst for palladium-catalyzed coupling reactions. Prominent applications include the Heck reaction, Suzuki coupling, Stille coupling, Sonogashira coupling, and Negishi coupling. These processes begin with two successive ligand dissociations followed by the oxidative addition of an aryl halide to the Pd(0) center:
...
Wikipedia

