Rhodium hexafluoride
 |
|
| Names | |
|---|---|
|
IUPAC name
rhodium(VI) fluoride
|
|
| Other names
rhodium hexafluoride
|
|
| Identifiers | |
|
3D model (JSmol)
|
|
|
|
|
|
| Properties | |
| F6Rh | |
| Molar mass | 216.91 g/mol |
| Appearance | black crystalline solid |
| Density | 3.71g/mL |
| Melting point | ≈ 70 °C (158 °F; 343 K) |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
| Infobox references | |
Rhodium hexafluoride, also rhodium(VI) fluoride, (RhF6) is the inorganic compound of rhodium and fluorine. A black volatile solid, it is a highly reactive material, and a rare example of a rhodium(VI) compound.
Rhodium hexafluoride is prepared by reaction of rhodium metal with an excess of elemental fluorine:
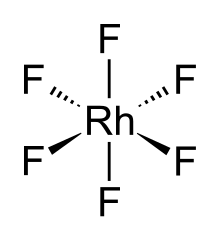
The RhF6 molecule has octahedral molecular geometry. Consistent with its d3 configuration, the six Rh–F bond lengths are equivalent, being 1.824 Å. It crystallises in an orthorhombic space group Pnma with lattice parameters of a = 9.323 Å, b = 8.474 Å, and c = 4.910 Å.
Like some other metal fluorides, RhF6 is highly oxidising. It attacks even glass even in the absence of water. It can even react with elemental oxygen.
...
Wikipedia
