Retinal edema
| Macular edema | |
|---|---|
 |
|
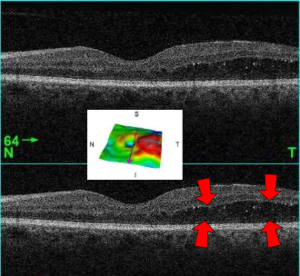
| A 61-year-old man with medical history of type 2 diabetes that presents a macular edema, evidenced by an OCT (the edema marked with arrows). The central image is a 3D reconstruction of the retinal thickness (the edema is coloured in red). | |
| Classification and external resources | |
| Specialty | ophthalmology |
| ICD-10 | H35.8 |
| ICD-9-CM | 362.01, 362.53 |
Macular edema occurs when fluid and protein deposits collect on or under the macula of the eye (a yellow central area of the retina) and causes it to thicken and swell (edema). The swelling may distort a person's central vision, because the macula holds tightly packed cones that provide sharp, clear, central vision to enable a person to see detail, form, and color that is directly in the centre of the field of view.
The causes of macular edema are numerous and different causes may be inter-related.
Cystoid macular edema (CME) involves fluid accumulation in the outer plexiform layer secondary to abnormal perifoveal retinal capillary permeability. The edema is termed "cystoid" as it appears cystic; however, lacking an epithelial coating, it is not truly cystic. The cause for CME can be remembered with the mnemonic "DEPRIVEN" (diabetes, epinepherine, pars planitis, retinitis pigmentosa, Irvine-Gass syndrome, venous occlusion, E2-prostaglandin analogues, nicotinic acid/niacin).
Diabetic macular edema (DME) is similarly caused by leaking macular capillaries. DME is the most common cause of visual loss in both proliferative, and non-proliferative diabetic retinopathy.
Macular edema sometimes occurs for a few days or weeks after cataract surgery, but most such cases can be successfully treated with NSAID or cortisone eye drops. Prophylactic use of Nonsteroidal anti-inflammatory drugs has been reported to reduce the risk of macular edema to some extent.
In 2010 the US FDA approved the use of Lucentis intravitreal injections for macular edema.
Iluvien, a sustained release intravitreal implant developed by Alimera Sciences, has been approved in Austria, Portugal and the U.K. for the treatment of vision impairment associated with chronic diabetic macular edema (DME) considered insufficiently responsive to available therapies. Additional EU country approvals are anticipated.
...
Wikipedia
