Repinotan
 |
|
| Clinical data | |
|---|---|
| Routes of administration |
Oral |
| Legal status | |
| Legal status |
|
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
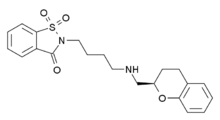
| Formula | C21H24N2O4S |
| Molar mass | 400.491 g/mol |
| 3D model (Jmol) | |
|
|
|
|
|
|
|
Repinotan (BAYx3702), an aminomethylchroman derivative, is a selective 5-HT1A receptor full agonist with high potency and efficacy. It has neuroprotective effects in animal studies, and was trialed in humans for reducing brain injury following head trauma. It was subsequently trialed up to phase II for treatment of stroke, but while side effects were mild and consisted mainly of nausea, repinotan failed to demonstrate sufficient efficacy to justify further clinical trials. However, repinotan continues to be investigated for other applications, and was found to be effective at counteracting the respiratory depression produced by morphine, though with slight reduction in analgesic effects.
Bayer Healthcare AG (Wuppertal, Germany) first synthesized repinotan during early 2000s. As of 2004, it was expected to be filed by the NDA. Phase III trials for both ischemic stroke and traumatic brain injury were run in March 2002. However, these trials found the drug ineffective as a treatment (November, 2009).
In October 2010, further investigation showed repinotan able to counteract respiratory depression caused by morphine. Repinotan continues to be examined, but has not yet been commercially released.
Repinotan is an enantiomerically pure aminomethyl chromane derivative with a saccharinylbutyl substituent. It is classified as a synthetic organic and possesses five hydrogen bond acceptors and one hydrogen bond donor. Its topological polar surface area is 84.09 and it has seven rotatable bonds. In addition, its molecular weight is 400.15 g/mole. Repinotan has a formal charge of zero and a covalently-bonded unit count of one. It is similar in chemical and physical properties to the ligands quetiapine, PAT5A, and pioglitazone.
Bayer Healthcare AG synthesized Repinotan in three main reaction sequences. A form with a metabolically stable 14carbon-label was necessary for pharmacokinetic studies. The hydrochloride was hydroxylated in the 6-position of the chromane moiety.
For the first reaction sequence, [14C]-phenol was used as the starting compound. Michael adduct formation was then employed in the preparation of chromane rings with [14C]-phenol.
In the second reaction sequence, [Carbonyl-14C]-2-hydroxy-acetophenone was used first. Compound XII was formed from condensation of [carbonyl-14C-]-2-hydroxy-acetophenone with dimethyl oxylate and ring closure. Hydrogenolytical debenzylation and hydrochloric acid created the final product.
...
Wikipedia
