Protoporphyrin IX
 |
|
| Identifiers | |
|---|---|
|
3D model (Jmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.008.213 |
|
PubChem CID
|
|
|
|
|
|
| Properties | |
| C34H34N4O4 | |
| Molar mass | 562.658 g/mol |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
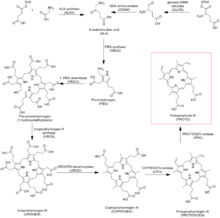
Protoporphyrin IX is an organic compound, which is one of the most common porphyrins in nature. It is a deeply colored pigment that is soluble in basic water. The free porphyrin is encountered in nature in the form of its iron complexes. When complexed with ferrous iron, the molecule is called heme. Hemes are prosthetic groups in some important proteins. These heme-containing proteins include hemoglobin, myoglobin, and cytochrome c. Complexes can also be formed with other metal ions, such as zinc.
Its biosynthesis is mediated by the enzyme protoporphyrinogen oxidase. Protoporphyrin IX is an important precursor to biologically essential prosthetic groups such as heme, cytochrome c, and chlorophylls. As a result, a number of organisms are able to synthesize this tetrapyrrole from basic precursors such as glycine and succinyl CoA, or glutamate. Despite the wide range of organisms that synthesize protoporphyrin IX the process is largely conserved from bacteria to mammals with a few distinct exceptions in higher plants.
In heme biosynthesis, the enzyme ferrochelatase converts it into heme b (i.e. Fe-protoporphyrin IX or protoheme IX).
In chlorophyll biosynthesis, the enzyme magnesium chelatase converts it into Mg-protoporphyrin IX.
Protoporphyrin IX reacts with iron salts in air to give the FeCl(PPIX).
...
Wikipedia