MDPV
 |
|
 |
|
| Clinical data | |
|---|---|
| Routes of administration |
Oral, insufflation, intravenous, rectal, vaporization |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Excretion | Primarily urine (renal) |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ECHA InfoCard | 100.222.786 |
| Chemical and physical data | |
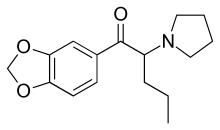
| Formula | C16H21NO3 |
| Molar mass | 275.343 g/mol (freebase) |
| 3D model (Jmol) | |
| Chirality | Racemic mixture |
|
|
|
|
|
|
|
Methylenedioxypyrovalerone (MDPV) is a stimulant of the cathinone class which acts as a norepinephrine-dopamine reuptake inhibitor (NDRI). It was first developed in the 1960s by a team at Boehringer Ingelheim. MDPV remained an obscure stimulant until around 2004 when it was reportedly sold as a designer drug. Products labeled as bath salts containing MDPV were previously sold as recreational drugs in gas stations and convenience stores in the United States, similar to the marketing for Spice and K2 as incense.
The hydrochloride salt exists as a very fine, hygroscopic, crystalline powder that tends to clump to itself, resembling something like powdered sugar. Its color can range from pure white to a yellowish-tan and has a slight odor that strengthens as it colors. Impurities are likely to consist of either pyrrolidine or alpha-dibrominated alkylphenones from either excess pyrrolidine or incomplete amination, respectively, during synthesis. These impurities likely account for its discoloration and fishy (pyrrolidine) or bromine-like odor, which worsens upon exposure to air, moisture, or bases.
Methylenedioxypyrovalerone has no record of FDA approved medical use. MDPV has been shown to produce robust reinforcing effects and compulsive self-administration in rats, though this had already been provisionally established by a number of documented cases of misuse and addiction in humans, before the animal tests had been carried out.
MDPV is the 3,4-methylenedioxy ring-substituted analog of the compound pyrovalerone, developed in the 1960s, which has been used for the treatment of chronic fatigue and as an anorectic, but caused problems of abuse and dependence.
...
Wikipedia
