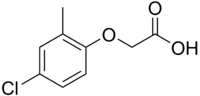
MCPA
 |
|
 |
|
| Names | |
|---|---|
|
IUPAC name
(4-Chloro-2-methylphenoxy)acetic acid
|
|
| Other names
4-Chloro-o-tolyloxyacetic acid
MCPA |
|
| Identifiers | |
|
94-74-6 |
|
| 3D model (Jmol) | Interactive image |
| ChEBI |
CHEBI:50099 |
| ChEMBL |
ChEMBL394657 |
| ChemSpider |
6935 |
| ECHA InfoCard | 100.002.146 |
| KEGG |
C18528 |
| PubChem | 7204 |
|
|
|
|
| Properties | |
| C9H9ClO3 | |
| Molar mass | 200.62 g·mol−1 |
| Appearance | White to light brown solid |
| Density | 1.18-1.21 g/cm3 |
| Melting point | 114 to 118 °C (237 to 244 °F; 387 to 391 K) |
| 825 mg/L (23 °C), amine salt: 866 g/L ester: 5 mg/L |
|
| Hazards | |
| Safety data sheet | External MSDS |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
MCPA (2-methyl-4-chlorophenoxyacetic acid) is a powerful, selective, widely used phenoxy herbicide. The pure compound is a brown-colored powder.
In 1936 investigations began at ICIs Jealott's Hill research center into the effects of the plant hormones auxins on plant growth looking specifically for a way to kill weeds without harming crops such as corn.
Synthesis of MCPA was first reported by Synerholme and Zimmerman in 1945 and by Templeman and Foster in 1946. Templeman and Foster were searching for compounds with similar or greater selective activity than 1-naphthaleneacetic acid in inhibiting the growth of weeds while not adversely affecting the growth of cereal grains. They synthesized MCPA from the corresponding phenol by exposing it to chloroacetic acid and dilute base in a straightforward substitution reaction:
MCPA is used as an herbicide, generally as its salt or esterified forms. Used thus, it controls broadleaf weeds, including thistle and dock, in cereal crops and pasture. It is selective for plants with broad leaves, and this includes most deciduous trees. Clovers are tolerant at moderate application levels. It is currently classified as a restricted use pesticide in the United States. Its toxicity and biodegradation are topics of current research. One formulation is described by its manufacturer as "designed for specific markets that require the safest possible phenoxy product, primarily for use in the Pacific Northwest". Though not extremely toxic, it has recently been determined that MCPA can form complexes with metal ions and thereby increase their bioavailability, though there is also work being done to utilize this ability.
Because it is inexpensive, MCPA is used in various chemical applications. Its carboxylic acid group allows the formation of conjugated complexes with metals (see above). The acid functionality makes MCPA a versatile synthetic intermediate for more complex derivatives.
...
Wikipedia
