L27 domain
| L27_1 |
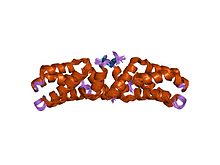
hetero-tetrameric l27 (lin-2, lin-7) domain complexes as organization platforms of supra-molecular assemblies
|
| Identifiers |
| Symbol |
L27_1 |
| Pfam |
PF09058 |
| InterPro |
IPR015143 |
|
|
| L27_2 |

2.1 angstrom crystal structure of the pals-1-l27n and patj l27 heterodimer complex
|
| Identifiers |
| Symbol |
L27_2 |
| Pfam |
PF09045 |
| InterPro |
IPR015132 |
|
|
| L27_N |

2.1 angstrom crystal structure of the pals-1-l27n and patj l27 heterodimer complex
|
| Identifiers |
| Symbol |
L27_N |
| Pfam |
PF09060 |
| InterPro |
IPR015145 |
|
|
The L27 domain is a protein domain that is found in receptor targeting proteins Lin-2 and Lin-7 (LIN7A, LIN7B, LIN7C), as well as some protein kinases and human MPP2 protein. The L27 domain is a protein interaction module that exists in a large family of scaffold proteins, functioning as an organisation centre of large protein assemblies required for the establishment and maintenance of cell polarity. L27 domains form specific heterotetrameric complexes, in which each domain contains three alpha-helices. The L27_2 domain is a protein-protein interaction domain capable of organising scaffold proteins into supramolecular assemblies by formation of heteromeric L27_2 domain complexes. L27_2 domain-mediated protein assemblies have been shown to play essential roles in cellular processes including asymmetric cell division, establishment and maintenance of cell polarity, and clustering of receptors and ion channels. Members of this family form specific heterotetrameric complexes, in which each domain contains three alpha-helices. The two N-terminal helices of each L27_2 domain pack together to form a tight, four-helix bundle in the heterodimer, whilst the third helix of each L27_2 domain forms another four-helix bundle that assembles the two units of the heterodimer into a tetramer.
...
Wikipedia




