Hexazinone
 |
|
| Names | |
|---|---|
|
IUPAC name
3-Cyclohexyl-6-dimethylamino-1-methyl-1,3,5-triazine-2,4-dione
|
|
| Other names
Velpar
Hexazinone |
|
| Identifiers | |
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.051.869 |
| KEGG | |
|
PubChem CID
|
|
|
|
|
|
| Properties | |
| C12H20N4O2 | |
| Molar mass | 252.31 |
| Appearance | White crystalline solid |
| Density | 1.25 g/cm3 |
| Melting point | 116 °C (241 °F; 389 K) |
| Soluble | |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Hexazinone is an organic compound that is used as a broad spectrum herbicide. It is a colorless solid. It exhibits some solubility in water but is highly soluble in most organic solvents except alkanes. A member of the triazine class herbicides, it is manufactured by DuPont and sold under the trade name Velpar.
It functions by inhibiting photosynthesis and thus is a nonselective herbicide. It is used to control grasses, broadleaf, and woody plants. Approximately 33% is used on alfalfa, 31% in forestry, 29% in industrial areas, 4% on rangeland and pastures, and < 2% on sugarcane.
Hexazinone is a pervasive groundwater contaminant, due to its high water solubility
Hexazinone is widely used as a herbicide. It is a non-selective herbicide from the triazine family. It’s is used among a broad range of places. It is used to control weeds within all sort of applications. From sugarcane plantations, forestry field nurseries, pineapple plantations to high- and railway grasses and industrial plant sites.
Hexazinone was first registered in 1975 for the overall control of weeds and later for uses in crops.
Triazines like hexazinone can bind to the D-1 quinone protein of the electron transport chain in photosystem II to inhibit the photosynthesis. These diverted electrons can thereby damage membranes and destroy cells.
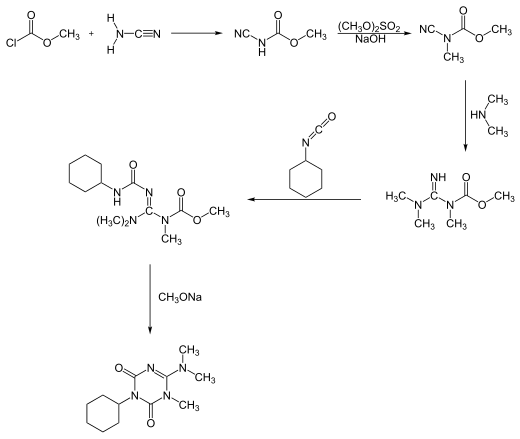
Hexazinone can be synthesized in two different reaction processes. One process starts with a reaction of methyl chloroformate with cyanamide, forming hexazinone after a five-step pathway:
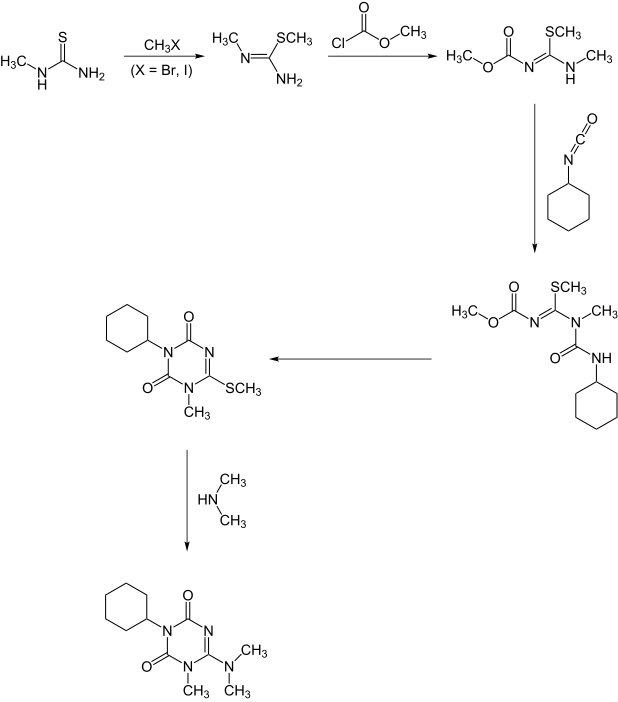
A second synthesis starts with methylthiourea.:
...
Wikipedia