Furazolidone
 |
|
 |
|
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration |
Oral-Local |
| ATC code | |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| ECHA InfoCard | 100.000.594 |
| Chemical and physical data | |
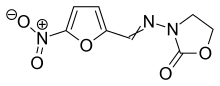
| Formula | C8H7N3O5 |
| Molar mass | 225.16 |
| 3D model (Jmol) | |
|
|
|
|
|
|
|
Furazolidone is a nitrofuran antibacterial agent. It is marketed by Roberts Laboratories under the brand name Furoxone and by GlaxoSmithKline as Dependal-M. Diafuron, medaron
Furazolidone has been used in human and veterinary medicine. It has a broad spectrum of activity being active against
In humans it has been used to treat diarrhoea and enteritis caused by bacteria or protozoan infections. It has been used to treat traveler's diarrhoea, cholera and bacteremic salmonellosis. Use in treating Helicobacter pylori infections has also been proposed.
Furazolidone is also used for giardiasis (due to Giardia lamblia), though it is not a first line treatment.
As a veterinary medicine, furazolidone has been used with some success to treat salmonids for Myxobolus cerebralis infections.
It has also been used in aquaculture.
Since furazolidone is a nitrofuran antibiotic, its use in food animals is currently prohibited by the FDA under the Animal Medicinal Drug Use Clarification Act, 1994.
It is used to differentiate micrococci and staphylococci.
It is believed to work by crosslinking of DNA.
Furazolidone is no longer available in the US. Though an effective antibiotic when all others fail, against extremely drug resistant infections, it has many side effects, and as with other nitrofurans generally, minimum inhibitory concentrations also produce systemic toxicity (tremors, convulsions, peripheral neuritis, gastrointestinal disturbances, depression of spermatogenesis.) Nitrofurans are recognized by FDA as mutagens/carcinogens, and can no longer be used since 1991.
...
Wikipedia
