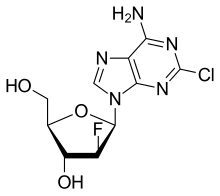
Clofarabine
 |
|
| Clinical data | |
|---|---|
| Trade names | Clolar, Evoltra |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607012 |
| Routes of administration |
Intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.159.663 |
| Chemical and physical data | |
| Formula | C10H11ClFN5O3 |
| Molar mass | 303.677 g/mol |
| 3D model (Jmol) | |
|
|
|
|
Clofarabine is a purine nucleoside antimetabolite marketed in the US and Canada as Clolar. In Europe and Australia/New Zealand the product is marketed under the name Evoltra. It is FDA-approved for treating relapsed or refractory acute lymphoblastic leukaemia (ALL) in children after at least two other types of treatment have failed. It is not known if it extends life expectancy. Some investigations of effectiveness in cases of acute myeloid leukaemia (AML) and juvenile myelomonocytic leukaemia (JMML) have been carried out. Ongoing trials are assessing its efficacy, if any, for managing other cancers.
Clolar was Food and Drug Administration (FDA) approved 28 December 2004. (Under accelerated approval regulations requiring further clinical studies.)
Clofarabine is a second-generation purine nucleoside analog designed to overcome biological limitations observed with ara-A and fludarabine. A 2´(S)-fluorine in clofarabine significantly increased the stability of the glycosidic bond in acidic solution and toward phosphorolytic cleavage as compared to fludarabine. A chlorine substitution at the 2-position of the adenine base avoids production of a 2-fluoroadenine analog, a precursor to the toxic 2-fluoro-adenosine-5´-triphosphate and prevents deamination of the base as compared to ara-A.
Clofarabine can be administered intravenously or given orally. Clofarabine enters cells via hENT1, hENT2, and hCNT2, where upon it is phosphorylated by deoxycytidine kinase to generate clofarabine-5´-monophosphate. The rate-limiting step in clofarabine metabolism is clofarabine-5´-diphosphosphate. Clofarabine-5´-triphosphate is the active-metabolite, and it inhibits ribonucleotide reductase, resulting in a decrease cellular dNTP concentrations, which promotes greater incorporation of clofarabine-5´-triphosphate during DNA synthesis. Embedded clofarabine-5´-monophosphate in the DNA promotes polymerase arrest at the replication fork, triggering DNA repair mechanisms that without repair lead to DNA strand breaks in vitro and cytochrome c-mediated apoptosis in vitro. Studies using cell lines have shown that clofarabine-5´-triphosphate can also be incorporated into RNA.
Mechanisms of resistance and turnover have been reported. Clofarabine-resistance arises from decreased deoxycytidine kinase activity in vitro. ABC transporter ABCG2 promotes export of clofarabine-5´-monophosphate and thus limits the cytotoxic effects of this analog in vivo. Biochemically, clofarabine-5’-triphosphate was shown to be substrate for SAMHD1, thus potentially limiting the amount of active compound in cells.
...
Wikipedia
