Caesium bromide
 |
|
| Names | |
|---|---|
|
IUPAC name
Caesium bromide
|
|
| Other names
Cesium bromide,
Caesium(I) bromide |
|
| Identifiers | |
|
3D model (Jmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.209 |
| EC Number | 232-130-0 |
|
PubChem CID
|
|
|
|
|
|
| Properties | |
| CsBr | |
| Molar mass | 212.809 g/mol |
| Appearance | White solid |
| Density | 4.43 g/cm3 |
| Melting point | 636 °C (1,177 °F; 909 K) |
| Boiling point | 1,300 °C (2,370 °F; 1,570 K) |
| 1230 g/L (25 °C) | |
| -67.2·10−6 cm3/mol | |
|
Refractive index (nD)
|
1.8047 (0.3 µm) 1.6974 (0.59 µm) 1.6861 (0.75 µm) 1.6784 (1 µm) 1.6678 (5 µm) 1.6439 (20 µm) |
| Structure | |
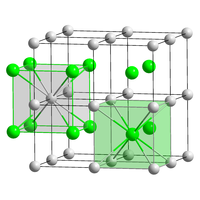
| CsCl, cP2 | |
| Pm3m, No. 221 | |
|
a = 0.4291 nm
|
|
|
Lattice volume (V)
|
0.0790 nm3 |
|
Formula units (Z)
|
1 |
| Cubic (Cs+) Cubic (Br−) |
|
| Hazards | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
|
LD50 (median dose)
|
1400 mg/kg (oral, rat) |
| Related compounds | |
|
Other anions
|
Caesium fluoride Caesium chloride Caesium iodide Caesium astatide |
|
Other cations
|
Sodium bromide Potassium bromide Rubidium bromide Francium bromide |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Caesium bromide is an ionic compound of caesium and bromine with the chemical formula CsBr. It is a white or transparent solid with a melting point a 636 °C that readily dissolves in water. Its bulk crystals have the cubic CsCl structure, but the structure changes to the rocksalt type in nanometer-thin film grown on mica, LiF, KBr or NaCl substrates.
Caesium bromide can be prepared via following reactions:
The direct synthesis is a vigorous reaction of caesium with other halogens. Due to its high cost, it is not used for preparation.
Caesium bromide is sometimes used in optics as a beamsplitter component in wide-band spectrophotometers.
...
Wikipedia
