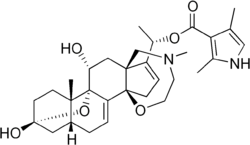
Batrachotoxin
 |
|
 |
|
| Identifiers | |
|---|---|
|
23509-16-2 |
|
| 3D model (Jmol) | Interactive image |
| ChemSpider |
10310314 |
| 2619 | |
| PubChem | 31958 |
|
|
|
|
| Properties | |
| C31H42N2O6 | |
| Molar mass | 538.68 g·mol−1 |
| Density | 1.304 g/mL |
| Hazards | |
| Main hazards | Highly toxic |
| Lethal dose or concentration (LD, LC): | |
|
LD50 (median dose)
|
2 μg/kg (mouse, sub-cutaneous) |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Batrachotoxin (BTX) is an extremely potent cardiotoxic and neurotoxic steroidal alkaloid found in certain species of frogs (poison dart frog), melyrid beetles, and birds (the pitohui, blue-capped ifrit, and little shrikethrush). Batrachotoxin was derived from the Greek word βάτραχος bátrachos "frog". Structurally-related chemical compounds are often referred to collectively as batrachotoxins.
It was named by scientists John W. Daly and Bernhard Witkop, who separated the potent toxic alkaloids fraction and determined its chemical properties. They isolated four major toxic steroidal alkaloids including batrachotoxin, isobatrachotoxin, pseudobatrachotoxin, and batrachotoxinin A. Due to the difficulty of handling such a potent toxin and the minuscule amount that could be collected, a comprehensive structure determination involved several difficulties. However, Takashi Tokuyama, who joined the investigation later, converted one of the congener compounds, Batrachotoxinin A, to a crystalline derivative and its unique steroidal structure was solved with x-ray diffraction techniques (1968). When the mass spectrum and NMR spectrum of batrachotoxin and the batrachotoxinin A derivatives were compared, it was realized that the two shared the same steroidal structure and that batrachotoxin was batrachotoxinin A with a single extra pyrrole moiety attached. In fact, batrachotoxin was able to be partially hydrolyzed using sodium hydroxide into a material with identical TLC and color reactions as batrachotoxinin A. The structure of batrachotoxin was established in 1969 through chemical recombination of both fragments. Batrachotoxinin A was synthesized by Michio Kurosu, Lawrence R. Marcin, Timothy J. Grinsteiner, and Yoshito Kishi in 1998.
According to experiments with rodents, batrachotoxin is one of the most potent alkaloids known: its subcutaneous LD50 in mice is 2 µg/kg. Meanwhile, its derivative, batrachotoxinin A, has a much lower toxicity with an LD50 of 1000 µg/kg.
...
Wikipedia
