Para-amino salicylic acid
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Paser |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration |
by mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 50–60% |
| Metabolism | liver |
| Excretion | kidney |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| ECHA InfoCard | 100.000.557 |
| Chemical and physical data | |
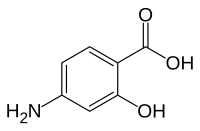
| Formula | C7H7NO3 |
| Molar mass | 153.135 g/mol |
| 3D model (JSmol) | |
| Melting point | 150.5 °C (302.9 °F) |
|
|
|
|
4-Aminosalicylic acid, also known as para-aminosalicylic acid (PAS) is an antibiotic primarily used to treat tuberculosis. Specifically it is used to treat active drug resistant tuberculosis together with other antituberculosis medications. It has also been used as a second line agent to sulfasalazine in people with inflammatory bowel disease such as ulcerative colitis and Crohn's disease. It is typically taken by mouth.
Common side effects include nausea, abdominal pain, and diarrhea. Other side effects may include liver inflammation and allergic reactions. It is not recommended in people with end stage kidney disease. While there does not appear to be harm with use during pregnancy it has not been well studied in this population. 4-Aminosalicylic acid is believed to work by blocking the ability of bacteria to make folic acid.
4-Aminosalicylic acid was first made in 1902 and came into medical use in 1943. It is on the World Health Organization's List of Essential Medicines, the most effective and safe medicines needed in a health system. As of 2005 a course of treatment for tuberculosis costs about 2,700 USD.
The main use for 4-aminosalicylic acid is for the treatment of tuberculosis infections.
Aminosalicylic acid was introduced to clinical use in 1944. It was the second antibiotic found to be effective in the treatment of tuberculosis, after streptomycin. PAS formed part of the standard treatment for tuberculosis prior to the introduction of rifampicin and pyrazinamide.
...
Wikipedia
