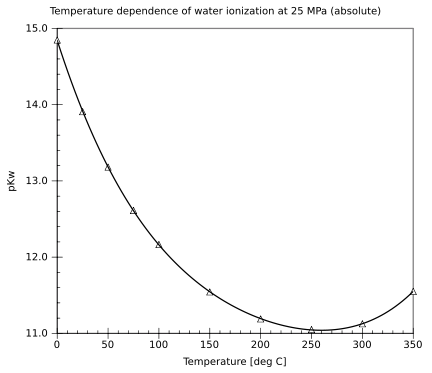
Water (data page)
This page provides supplementary data to the article properties of water.
Further comprehensive authoritative data can be found at the NIST Webbook page on thermophysical properties of fluids.
88.00 at 0 °C
86.04 at 5 °C
84.11 at 10 °C
82.22at 15 °C
80.36 at 20 °C
78.54 at 25 °C
76.75 at 30 °C
75.00 at 35 °C
73.28 at 40 °C
71.59 at 45 °C
69.94 at 50 °C
66.74 at 60 °C
63.68 at 70 °C
60.76 at 80 °C
57.98 at 90 °C
55.33 at 100 °C
Vapor pressure formula for steam in equilibrium with liquid water:
where P is equilibrium vapor pressure in kPa, and T is temperature in kelvins.
For T = 273 K to 333 K: A = 7.2326; B = 1750.286; C = 38.1.
For T = 333 K to 423 K: A = 7.0917; B = 1668.21; C = 45.1.
Data in the table above is given for water-steam equilibria at various temperatures over the entire temperature range at which liquid water can exist. Pressure of the equilibrium is given in the second column in kPa. The third column is the heat content of each gram of the liquid phase relative to water at 0 °C. The fourth column is the heat of vaporization of each gram of liquid that changes to vapor. The fifth column is the PV work done by each gram of liquid that changes to vapor. The sixth column is the density of the vapor.
Data obtained from CRC Handbook of Chemistry and Physics 44th ed., p. 2390
‡Ice XI triple point is theoretical and has never been obtained
Note: ρ is density, n is refractive index at 589 nm and η is viscosity, all at 20 °C; Teq is the equilibrium temperature between two phases: ice/liquid solution for Teq < 0-0.1 and NaCl/liquid solution for Teq above 0.1 °C.
...
Wikipedia